A COMPREHENSIVE GUIDE TO TECHNICAL DIVE PLANNING / PART 2
07/10/2024
In the previous part of the article we have discussed the event and the dive very broadly. In the second part of the article we will focus on planning the dive itself. That is, everything you need to plan for staying safely underwater and returning to the surface.

Decompression
It will soon be 120 years since the publication of the first Haldane decompression tables and we are still not very clear on the issues that are most typical for diving activity. According to recent research, it seems that algorithms generating deep stops are out of date and the best we have is the Bühlmann model modified by gradient factors. The choice of the gradient factor is already the first step in decomposition planning. Current trends show that the safest and most effective is to use a GF low in the range of 40 - 50 and a GF high of 60 - 80. What you set in your planning app and your dive computer will affect the depth of the first stop (GF low) and the level of tissue saturation on surfacing (GF high).

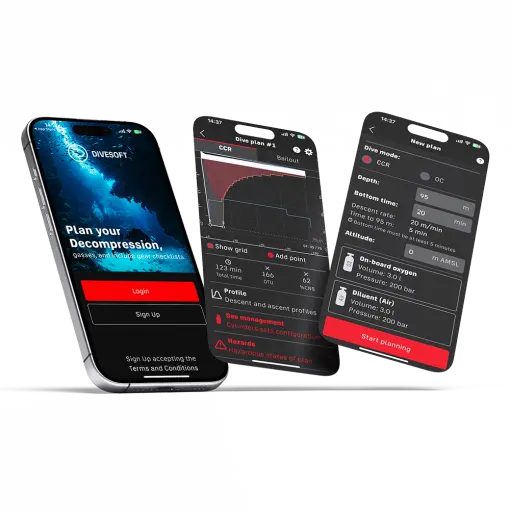
Speaking of scheduling apps, that's the current modern trend. After the use of spreadsheets and planning software on the desktop computer comes mobile phones, which are now more powerful than PCs in the days of the first decompression software. Mobile phones are always at our fingertips and their operation is easy. Divesoft.app has an advanced scheduler that is continually improved. You choose whether you plan for open or closed circuit, set all the parameters and enter the dive profile, i.e. maximum depth and time. The app takes into account the subtle nuances of personal customisation of the planner. It is also possible to customize the profile for multi-level dives by adding additional points to the chart. This makes it suitable for advanced cave dives with multi-level depth profiles.
A bailout plan is available in CCR mode. This is a must for planning with a rebreather, because decompression on the open circuit at a moment at which you can no longer dive with CCR due to a failure, is usually longer and you consume a significant amount of gas. You can further adjust the plan to make it acceptable and feasible in the planned conditions.

Once you are satisfied, you can transfer the plan to a .pdf format using the Print function and print the plan by sending it to the printer. You can take this plan underwater with you and follow it.
In any case, every technical diver should have an understanding of the ratio of time to surface at what depth and be able to derive the decompression time off the top of their head. Some people use this as a basic method and software just to confirm, but it can work the other way around. In any case, it comes in handy when the decompression computer fails.
Gas management
The amount of time we can spend at the bottom is determined by the amount of gas we have available. Everything can be planned by calculations on paper or by entering it into a dive planning app, where it is easier and infinitely faster.

When planning an open circuit dive, we have to take into account that we always have to get to the surface safely even if we or a team member runs out of gas at the worst possible moment, i.e. at the end of the planned time on the bottom. Above all, we must know the minimum amount of gas we need to surface. We take into account that we will be breathing two from one apparatus and under stress, so we have to add our partner's consumption to our own and multiply it by the stress factor of 1.5. We must not forget to multiply the result by the ambient pressure. We calculate the output until the first gas switch, where the partner switches to decompression gas and therefore has their supply. We can be conservative and calculate the pressure of the maximum depth, or we can be more realistic and calculate the average pressure between the maximum depth and the depth of the first gas switch. Let's take the example of a dive to 60 m with a gas switch at 21 m. The pressure at 60 m is 7 atm, at 20 m it is 3 atm. The average pressure will therefore be (7+3) / 2 = 5 atm.
My consumption is 15 l/min.
Partner's consumption 15 l/min
Stress coefficient 1.5 ×
Calculation: (15 + 15) × 1.5 × 5 = 225 l/min
Ascent time from 60 m to 21 m at 10 m/min = 4 minutes
Calculation: 4 × 225 = 900 litres.
On the bottom, however, we'll probably deal with a crisis situation and shoot a buoy to the surface, for example. For this reason it is a good idea to allow some extra time, at least 3 minutes. That's 3 × (15 l/min + 15 l/min) × 1.5 × 7 atm = 945 litres
900 litres is the amount of gas we will need to ascend to 21 metres, where we will switch to decompression gas, and 945 litres are needed to deal with the situation at the bottom, so 1845 litres in total. If we have 2×12 litre cylinders, that's 77 bar in the cylinder that we have to start surfacing with. 123 bar is therefore available for the dive itself, which is enough for us at 60 metres:
(123 bar × 24 l) / (15 l/min × 7 atm) = 28 minutes
We count decompression cylinders separately. Here we calculate the simple consumption.
Gas management for CCR
In the case of closed circuit diving, gas management is simple for CCR and the bailout bottle remains unused.
Oxygen consumption is simply the dive time × 1.5 l/min. Although the physiological consumption is lower, we have to calculate the gas losses associated with the dive.

The diluent consumption depends on the loop volume and the depth of immersion, or repeated descents. If the profile is simple, we can calculate the loop volume multiplied by the pressure. For a more realistic result, we multiply by two to account for gas losses during small changes in depth.
More challenging and critically important is gas management planning for the bailout. We have to take into account that a bailout will happen at the worst moment of the dive, i.e. at the end of the planned time on the bottom at maximum depth. We add again 3 minutes to solve the problem.
The question is what consumption to choose. Since this is a really critical situation where we have to switch from CCR to OC and CO2 hit is in play, we calculate a consumption of 50 l/min. This applies for at least the first 10 minutes of the rescue plan.
So we count the first 3 minutes on the bottom, let's say again at 60 meters:
3 × (50 l/min × 7 atm) = 1050 l
Ascent to 21 m takes 4 minutes and the average pressure is 5 atm: 4 × (50 l/min × 5 atm) = 1000 l
To climb to 21 metres, we need 2050 litres, which is equivalent to one Al80 bottle.
Next, we have 3 minutes left at a consumption of 50 l/min and the rest is calculated with a consumption of 25 l/min. This is a realistic calculation, as CCR divers easily lose their low consumption capabilities as they can breathe as much as they want with CCR, and their transition to OC is often accompanied by an unpleasant surprise at how much gas they have exhaled.
Another interesting element of the bailout plan is that we choose a conservative PO2 value on the bottom with CCR, and the subsequent decompression is actually longer than if the same dive had been done with OC.
Calculating the consumption at all decompression stops can be rather tedious, so it is more convenient to use a scheduling application. Divesoft.app assumes that the diver is using a stock of bailout gases that they do not change, so the app has a virtual stock of cylinders that the user uses most often. He then chooses from these cylinders when planning the dive, but of course he can add or change any other mixture during the planning process. In the application settings, you can also set the length of time to solve a problem, the consumption in a critical (stressful) situation, the length of a stressful situation and the consumption outside a stressful situation. The app then calculates everything for you.
Selecting Mixtures and Setpoints
Choosing a mixture is always like walking a tightrope. We have to balance over dangerous precipices, and we don't want to fall into any of them. Those chasms have names like decompression sickness, oxygen poisoning, nitrogen anesthesia and gas density.
The first question should be: how much oxygen? The answer might be as much as possible, which is partly true. We metabolize oxygen, so it doesn't contribute to decompression sickness (quite the opposite, in fact), so the more we have in the mix, the less inert gases we'll suck into our tissues, and the less decompression duties we'll have.
On the other hand, there must not be too much of it, lest we endanger ourselves with oxygen poisoning. We must carefully consider the exposure time to determine the maximum partial pressure of oxygen (PO2 ). The NOAA table of CNS oxygen exposures will help us to do this. This table is not very exact, and we suspect that many things are a little different, but it is the best we have for now.
PO2 (bar) / Single dive exposure (minutes) / 24-hour exposure (minutes)
1,6 / 045 / 150
1,5 / 120 / 180
1,4 / 150 / 180
1,3 / 180 / 210
1,2 / 210 / 240
1,1 / 240 / 270
1,0 / 300 / 300
0,9 / 360 / 360
0,8 / 450 / 450
0,7 / 570 / 570
0,6 / 720 / 720
In the case of an open circuit, maximum PO2 for the mixture at maximum depth and PO2 for decompression. This partial pressure will only ever be reached at maximum depth and will decrease with decreasing depth. With the circuit closed, however, the partial pressure remains the same all the time (until we switch to a different setpoint), in which case the oxygen exposure is much more severe. Hence, with a rebreather we always choose much more conservative values. While with OC we can expect a PO2 of 1.4 on the bottom and 1.6 on decompression, with CCR the maximum PO2 will be 1.2 on the bottom and if the total dive length allows it, we can get up to 1.4 on decompression. Of course we are talking about the preset setpoint. The actual mixture should be 0.2 bar PO2 lower still at the target depth so that we have enough reserve to dilute the loop in case of trouble.
So how do we find out how much oxygen should be in the mixture? By a simple calculation of Dalton's law. We calculate the absolute pressure at the maximum depth.
F = PO2 / P
For example, for a depth of 60 m and a selected partial pressure of 1.4, calculate the oxygen fraction as follows:
F = 1,4 / 7
F = 0,2
We then find that for a depth of 60 m we use 20% oxygen in the mixture. For closed circuit diving, the pattern remains the same of course, but we have different entry criteria. In particular, the maximum PO2 will be 1.2 bar. But we also have to ensure that the total gas at depth is dilutable. It is therefore advisable to select a maximum PO2 of the mixture that is 0.2 bar lower than the setpoint. In our case, 1 bar.
F = PO2 / P
F = 1 / 7
F = 0,14
The proportion of oxygen in the diluent will be 14%.
The CNS oxygen toxicity breaks down by 50% every 90 minutes and this should be taken into account when planning repeat dives.
In addition to CNS oxygen toxicity, we have whole-body toxicity arising from prolonged exposures even at lower POs2 . We calculate it in OTU (oxygen toxicity unit). One unit of OTU is produced by a minute exposure to PO2 1.0. A daily limit of 300 OTUs is set for whole body toxicity. If we were calculating the entire OC dive, we would have to take the individual levels at each PO2 level and add them together.
For example, a dive to 60m for 20 minutes on a mixture with 20% oxygen and decompression to EAN 50 from a depth of 21m. We already know that the PO2 at the bottom is 1.4 bar
OTU bottom 1,4 × 20 = 28
OTU output (average pressure 5 bar) (5 × 0.2) × 4 = 4
OTU deco 21m 1,5 × 4 = 6
OTU deco 18 m 1,4 × 2 = 2,8
OTU deco 15 m 1,2 × 1 = 1,2
OTU deco 12 m 1,1 × 3 = 3,3
OTU deco 9 m 0,9 × 5 = 4,5
OTU deco 6 m 0,8 × 22 = 17,6
Total OTUs = 67.4
It is clear from the previous calculation that whole body toxicity does not play a very significant role in open circuit diving. However, it is different in rebreather diving. If we perform the same dive with CCR, the OTU calculation will obviously be much easier, and let's see what it does to the result. Let's take a setpoint of 1.2 bar on the bottom and 1.4 on the decompression.
The time at setpoint 1.2 is 20 minutes and according to the decompression planner the total output at setpoint 1.4 is 39 minutes.
OTU bottom 1,2 × 20 = 24
OTU decompression = 1.4 × 39 = 54.6
Total OTUs per dive = 78.6
We can see that a separate dive won't affect us much. The problem may arise in the case of repeated dives. When diving with a CCR at an attractive site or expedition, where several dives a day for a week or more are dived, the whole body form of oxygen toxicity may already be a problem that must be seriously taken into account.

Clean head
Once we have our oxygen, we then need to focus on making sure that at each dive depth our cognitive, mental and reactionary abilities are as little affected as possible by the narcotic effect of inert gases at high pressure. If we are diving with air on an open circuit, we can say that at a depth of 30 meters our mind is still acceptably efficient. So we will take this depth and relate our calculations to it. We will call this depth the equivalent narcotic depth (END).
So we need to find out what the partial pressure of nitrogen is at those 30 meters with air:
PN2 = FN2 × P
PN2 = 0,78 × 4
PN2 = 3,12 bar
To be even more conservative, we can use the simplified 3 bar PN2, which corresponds to a depth of 28 m.
If we want to find the amount of helium in the mixture, we can actually add the sum of the maximum partial pressures of oxygen and nitrogen, and subtract it from the ambient pressure at the maximum depth. So if we use the 60 meter depth from the previous examples, it will look like this for planning the OC of the mixture:
PHemax = P - (PO2 + PN2)
PHemax = 7 - (1,4 + 3)
PHemax = 2,6 bar
According to the already known formula we calculate the fraction of helium
FHe = 2.6 / 7 = 0.37
If we take all the information together, we get a trimix 20/37.If we wanted to use a standardized mixture, we would go for the closest one, which is trimix 18/45.
On the other hand, if we add too much helium and still use helium penalties, which I don't want to go into, the decompression software will unnecessarily increase the decompression time.
If we use the same procedure to calculate the mixture for the diluent, where we want a maximum oxygen partial pressure of 1 bar, we arrive at a mixture of trimix 14/43.

Rescue and evacuation plan
Last, but certainly not least, the most important element of planning is to think through the rescue and evacuation plan when things don't go as they should. We would prefer to tuck away the fact that a tragic or very dangerous event may happen as deeply as possible or even erase it altogether, but that could backfire badly. If we already have an advance plan of what to do at what time, what first aid, CPR or technical rescue equipment we will need from a given location, where to call in an emergency, where to take an injured or disabled DCS, it will make things significantly easier when something serious actually happens.
Make sure you have an adequate supply of oxygen and all CPR supplies in advance. If you can get an AED the better for you. Consider using a stretcher to get the casualty out of the water, scooters to get the casualty to the boat or shore.
Find out all safety contacts for paramedics, decompression chambers and insurance companies. Secure locations for arrival of rescue personnel, identify possible helicopter landing sites. There are many options to consider and it is up to each team to think carefully and prepare.

Conclusion
Planning a technical dive is a very complex matter and technical diving cannot be done without planning. If you dive the same sites repeatedly with the same or similar exposures, the plan can be used repeatedly and over time will become routine. However, one must never forget to update such a plan. The technical diver must be first and foremost.

Author: Jakub Šimánek
Divesoft.app
Plan your decompression, gasses, utilize checklists, check your logs + more!!
All for Free.